
* Apply- Resources and task in which students apply their knowledge gained during the lesson. * Explain- Embedded Google Slides™ presentation (includes lesson objectives, information slides, self-assessed questions). * Explore- Links to articles, video clips, virtual labs, and simulations for students to explore. **Please note: Kerboodle worksheets from scheme are not included due to license.** Note: Summary of the trend in reactivity The Net Enthalpy Changes (Thermodynamics) Activation Energies (Kinetics) Explaining the increase in reactivity down the group All of Group 1 elementslithium, sodium, potassium, rubidium and cesium react vigorously or even explosively with cold water. It can be used as a differentiated activity for the more able students within a group. The reactivity series is a hierarchical arrangement of elements based on their relative tendency to undergo chemical reactions. This could be used to follow up some work on the periodic table where the trends in reactivity in groups 1 and 7 have been identified. 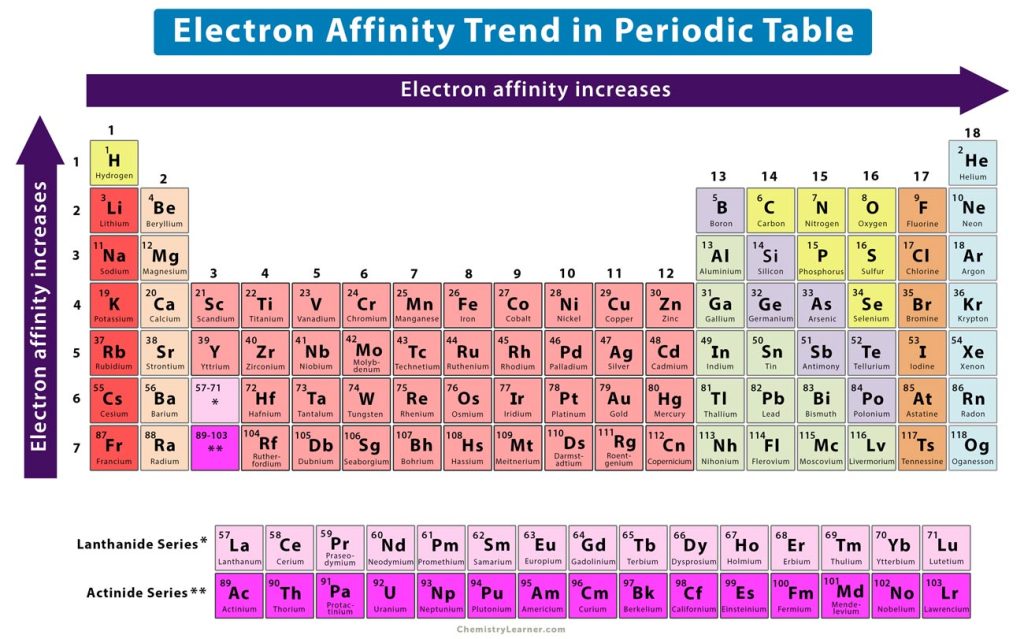 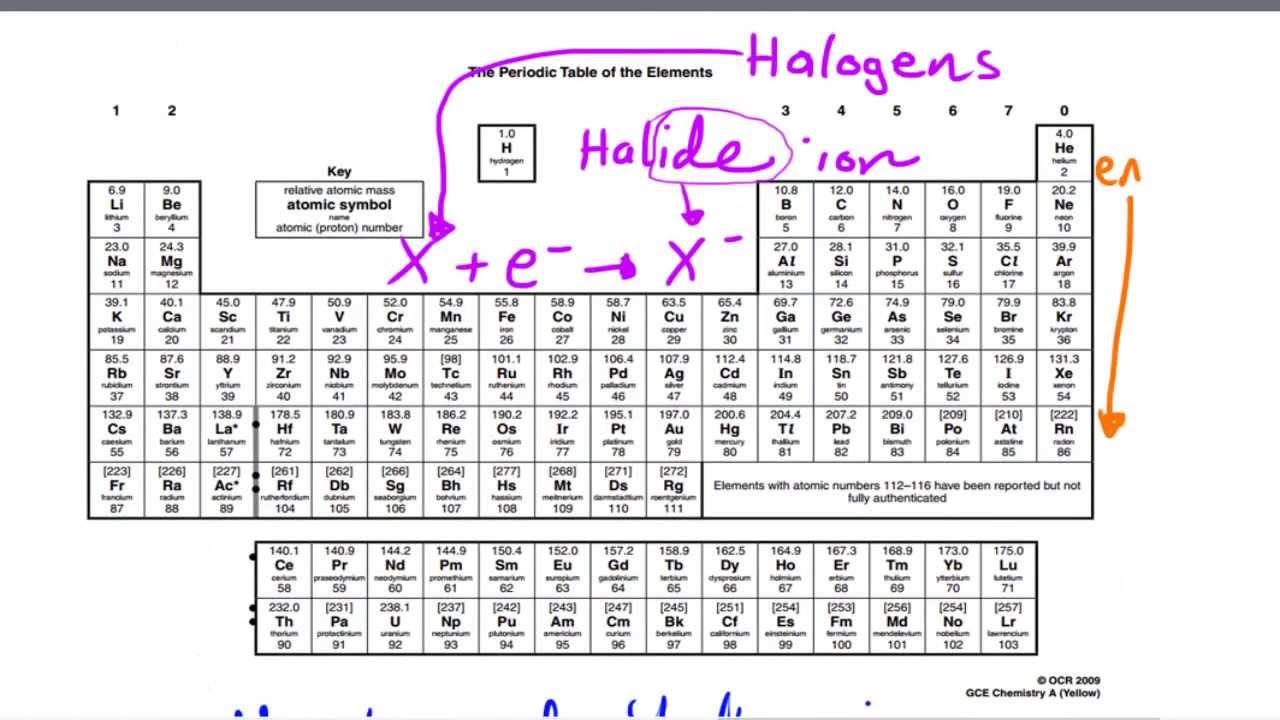
* Demonstrate understanding tasks (with answers) Chemistry for the gifted and talented: trends in reactivity in the periodic table. Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool to quickly predict an element's properties. **Īimed at a mixed ability class covering content 1-8. Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. Each of the elements found in this column, which are boxed in lavender in Figure 2.2.1 2.2. On moving from left to right horizontally in a periodic table, there is an increase in electronegativity. The Pauling scale is used to measure electronegativity. predict which element forms the most stable compounds in the +2 oxidation state. Electronegativity measures the ability of a molecule to gain electrons within a chemical bond. Based on the positions of the group 14 elements C, Si, Ge, Sn, and Pb in the periodic table and the general trends outlined in this section, classify these elements as metals, semimetals, or nonmetals. Students work through a variety of tasks to develop their understanding of the trends in reactivity in Group 1 and Group 7. Some periodic trends and chemical reactivity in the periodic table are as follows. As Z increases by a small interval, the shell number increases, and so the electrons in the valence shell are much farther from the nucleus and are more shielded by the electrons in the lower energy shells.**This lesson bundle covers the content for the AQA GCSE Chemistry C2-5 Explaining trends lesson in two formats- classroom based lesson presentation and digital worksheet Ideal for distance learning or covering missed content after absence. Only H-F forms strong hydrogen bonds and this is reflected in the boiling and melting points of. The stabilities of the hydrogen halides decrease down the group, but their acid strengths increase. Reactivity is a measure of how easily an element will combine with other elements to form compounds. The oxidizing ability of the halogens decreases markedly down the group: F 2 > Cl 2 > Br 2 > I 2, and only iodine is oxidized by nitric acid. 
\), we can see that when Z increases by one proton and the period increases (for example, going from Ne to Na), there is a relatively large decrease in Z eff. Learn about the trends in periodic table : Get detailed article on periodic table trends, overview, physical properties, chemical properties and examples.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
